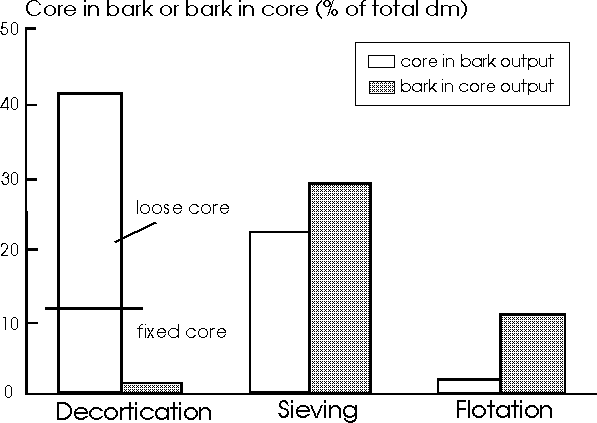
 Figure 1. Hemp component cross-contamination (as % mass of core dm in bast or bast dm in core), according to separation technique.
Figure 1. Hemp component cross-contamination (as % mass of core dm in bast or bast dm in core), according to separation technique.
New technology to harvest and store fibre hemp for paper pulp
Eddy A.A. de Maeyer 1 and Willem Huisman 2
1. Institute of Agricultural and Environmental Engineering (IMAG-DLO), P.O. Box 43, 6700 AA Wageningen, The Netherlands
2. Wageningen Agricultural University, Department of Agricultural Engineering and Physics, Bomenweg 4, 6703 HD Wageningen, The Netherlands
de Maeyer, E.A.A. and W. Huisman, 1994. New technology to harvest and store fibre hemp for paper pulp. Journal of the International Hemp Association 1: 38-41.
After mowing fibre hemp in September, field-drying is weather-dependent and therefore not reliable under Dutch weather conditions. Direct harvesting can be done by field choppers equipped with a row-independent header. Storage and preservation of the wet material on the farm is possible under acid and alkaline anaerobic conditions. No losses of cellulose were measured, but fibre strength was decreased. Bark and core can easily be separated by water flotation of the preserved chopped stems. Decortication of the stems in the field was less effective and would be more expensive.
In 1990, the Dutch government initiated a four year programme to research the technical and economic feasibility of cultivating fibre hemp for paper pulp. Both bast and core had to be processed and the objective was to cultivate a crop with a maximum fibre yield. This meant that harvest could not start before September and that traditional harvesting methods might not be suitable.
Fibre hemp is usually grown for textile fibres. For this application, the strength, colour, softness and fineness of the bast fibres are very important and harvesting technique focuses on these aspects (for example, a retting process is necessary). Because of the decreased demand for bast fibres, in most areas the mechanization of bast fibre crops is not advanced. In the Ukraine, hemp is mown and laid down in swaths to ret (van der Werf 1994). In the USA, an experimental machine was developed to cut kenaf. The stems were also laid down in a swath to ret or to dry (USDA 1988). In Australia, sugar cane harvesters and field choppers have been used to harvest kenaf for paper pulp (Wilkes et al. 1969, Wood et al. 1978). Both types of machines technically functioned well, but the harvest costs of a sugar cane harvester were found to be much higher than those of a field chopper. In the USA and Australia, initial attempts to start a kenaf-processing industry failed.
In France, 6000 ha of fibre hemp is grown for processing into paper pulp. Two harvesting systems are used, depending on whether the seed is to be harvested or not (FNPC 1985). For the best and highest fibre yield, the hemp is cut with a mower conditioner for field drying in the third week of August. In about 4 days, the moisture content is under 15% and the hemp is baled with a round baler. However, if the seed is also to be harvested, the stem tops are cut and threshed with a combine in the first week of September. Subsequently, the crop is mown and laid down in a swath to dry. With this second harvesting system the fibre yield is much lower (depending on the cutting height of the combine) and because of poor weather conditions in September, the fibre quality can be lower.
From the beginning of the Dutch Hemp Research Programme, two specific research topics were formulated. First, a system for preservation and storage of the harvest was needed so as to enable supply of pulp processing plants with fibres for the whole year. Because hemp may not be harvested dry, many experiments were performed concerning the preservation and storage of wet hemp. Secondly, the processing of hemp into paper pulp demands a separation of bark and core because these fractions differ physically (fibre length and diameter) and chemically (cellulose and lignin content) (Zomers et al. 1991). The separation may be done as a harvest operation in the field or as a preprocessing step of the pulping. The Institute of Agricultural and Environmental Engineering (IMAG-DLO), together with the Department of Agricultural Engineering and Physics of the Wageningen Agricultural University, investigated these aspects to determine the best processing chain in which harvest, preservation, storage and bark/core separation are integrated.
Field tests were carried out with a cutter bar, a mower conditioner, balers, field choppers and a decorticator. All machines, except the decorticator, are commonly used in other crops. When relevant and necessary, minor adaptions were made to make them suitable for hemp. The main objective of the field tests was to learn and solve the difficulties in handling a fibre hemp crop utilizing different techniques.
We investigated the possibility of wet preservation by anaerobic storage. The hemp stems were cut in short pieces (1 cm) and preserved in bottles of 1.5 l capacity. After flushing with nitrogen, the bottles were closed air tight. To create different environments, we used the following additives: Foraform (Barenbrug Holland BV), a silage additive consisting primarily (92%) of formic acid; kitchen salt; sodium hydroxide; and Silage inoculant 1188 (Pioneer Hi-bred Nederland BV), a silage additive containing Lactobacillus plantarum and Streptococcus facium. After preservation, pH was measured of a 1:10 aquatic extract. The cellulose and hemicellulose sugars and the lignin were determined using an analysis derived from TAPPI-249 and TAPPI-222 (de Maeyer and Huisman 1994). Bast fibres were cooked for 1 hour in a 2% sodium hydroxide solution and the strength was measured in a controlled environment (23o C, relative humidity 50%) with a stelometer according to ASTM D-1445 (ASTM 1967).
We used 3 methods to separate bark and core: decortication of green stems in the field, sieving of chopped stems, and flotation of chopped stems. The decorticator broke the core of whole stems and beat the core pieces from the bark. The sieving and flotation of chopped stems were done after a 6 month wet preservation period. The sieve dimensions were 2 x 4 cm. With flotation, the hemp was thrown into a bath of slow moving water in which part of the pieces floated and the rest sank. By measuring bark and core masses, we determined the efficiency of each operation.
For storage of dry hemp, the moisture content must be less than 15% to avoid fibre decay by micro-organisms. In France, field drying takes at least 4 days for the crop to reach the necessary dry matter (dm) content of 85%. Comparing weather data from two Dutch and two French weather stations, we estimate that in the Netherlands, on average, 6 days of dry weather in succession are needed to have the hemp sufficiently dried. Based upon data of the workability of the grain harvest (Portiek 1975), in September for 9 out of 10 years, only 3.5 days are available to harvest winter wheat with a moisture content below 24%. If the moisture content of wheat is higher than 23%, it is not expected that a mown hemp crop, lying on the ground, will reach the necessary moisture content of less than 15%. So, in September, the field drying period would be longer than 6 days for many of the years and the crop would start dew retting. This would decrease bast fibre strength (Hessler 1945, Robinson 1946). If the crop stays wet, retting can even become rotting. So, we concluded that field drying is not reliable in the Netherlands in September.
Chopping of the standing crop is less weather dependent and a field chopper with a row independent header can mow the crop easily. Fewer problems occurred with a drum type chopper than with a flywheel type chopper. To avoid blockages, the crop chute must be smooth and without any augers or blind areas. The knives must be sharp and the chopping length set short (6 mm). To handle hemp, great attention has to be paid to avoid bark wrapping around rotating machine parts.
After chopping, the hemp has a dm content of 33%. Artificial drying is too expensive, so we tried to preserve the hemp by ensiling (wet anaerobic preservation). In corn or grass silages, lactic acid bacteria consume available sugars and produce lactic acid to yield a pH of 4.2. Then the product is preserved and no further losses occur (McDonald et al. 1991). In hemp silage, the pH did not drop to 4.2, even after addition of lactic acid bacteria. The reason for this was lack of the necessary sugars. Only 4 g of sugars per kg of stem dm were available and based upon grass silage, about 75 g are needed for a sufficient fermentation (Wieringa 1961). Despite the inadequate fermentation, no cellulose or hemicellulose was lost in silage (Table 1). Only with the addition of sodium hydroxide did some hemicellulose and lignin dissolve. This was also shown by de Groot et al. (1994). Impregnation of hemp core, during 30 minutes with 1 M sodium hydroxide at 20o C, removed some xylan (the main component of the hemicellulose) and some lignin.
It is important to maintain fibre strength when processing the bast for paper pulp. Paper tensile strength is positively correlated with the quantity and the quality of the bast fibres. The bast fibre strength was best preserved in an acidic (pH<4.5) or alkaline (pH>12) environment (Table 2). However, even under these circumstances fibre strength decreased. Because the control fibres, after 3 and 6 months, were not the same (dry compared to frozen fibres), no clear results can be given on the decrease of fibre stength in time. However, the big difference between the data after 3 and 6 months' storage indicate that fibre strength may be slowly decreasing, even in a stable acidic or alkaline environment. More research is necessary to determine how much this strength decreases and whether this decrease can be stopped.
Table 1. pH of ensiled hemp and composition of dry stored and ensiled hemp
Preservation Additive Dosage pH Cellulose Hemicellulose Lignin
[g.kg-1 of fresh [%] [%] [%]
cut hemp]
Dry 47.1a1 14.0a 15.3a
Silage None 5.6 49.0a 13.4a 15.4a
Foraform 20 3.7 48.7a 13.6a 15.2a
Kitchen salt 30 5.4 47.2a 13.3a 14.8a
Sodium hydroxide 30 12.6 49.6a 11.2b 11.8b
Silage inoculant 1188 106 bacteria 5.1 48.1a 12.8ab 14.6a
1Within each column, means with the same letter are not significantly different at P<0.05 by the LSD test.
Table 2. Fibre strength [g/tex] of dry, frozen and wet anaerobically preserved bark.
Preservation Preservation period
environment
3 months 6 months
Dry 54.1a1
Frozen 44.7a
Silage 3.5<pH<4.5 43.7b 29.9c
4.5<pH<5.5 38.1c 28.1cd
5.5<pH<6.5 39.0c 25.8d
pH>12 45.1b 35.1b
1Within each column, means with the same letter are not significantly different at P<0.05 by the LSD test.
Figure 1. Hemp component cross-contamination (as % mass of core dm in bast or bast dm in core), according to separation technique.
Figure 1 presents the results of decortication in the field and the separation of bark and core after chopping and ensiling, by sieving or by flotation. With decortication, the bark could not be cleaned well enough and still contained more than 40% of the core. About two thirds of this core was not attached to the bark. Bark and loose core should be easily separable by sieving since the size of the bark and of the loose core pieces greatly differ. The length of the core pieces ranged from 1 to 8 cm and the length of the bark from 0.5 to 2.5 m. The quantity of the remaining fixed core would be very difficult to reduce. It was clear that the quantity of fixed core mainly depended on stem diameter: the smaller the diameter, the higher the quantity of fixed core.
With field chopping, the bark was not cut as short as the core. The bark was very tough and the knives stripped some of the bark from the core. So after chopping, about 90% of the mixture consisted of clean bark and core pieces. This made it possible to separate bark and core by sieving or flotation. Sieving of chopped hemp resulted in a 'contamination rate' of both bark and core of about 25%, since the size difference between the chopped bark and core was not big enough to separate them by sieving. With flotation, very clean bast could be collected, although some bark floated because it stuck to a piece of core, resulting in its collection with the core.
A disadvantage of decortication is the low processing capacity. The costs would be high, approximately 110 Dfl per ton of dm, especially because the machine only can be used in hemp. Flotation seems most satisfactory and would cost approximately 5 Dfl per ton of dm. It is expected that a simple chopping operation of the floating material can reduce the mass of bark in the core.
Field choppers equipped with a row-independent header can harvest fibre hemp, but the sharpness of the knives and the throughput of the chopped fibres need special attention to prevent wrapping or blocking. The chopping operation loosens about 90% of the bark and core. Because the bark sinks in water and the core floats, flotation is an easy and effective separation method. This operation perfectly fits into a harvesting system with chopping and ensiling. In a system of cultivation, harvest and storage of fibre hemp, wet anaerobic preservation is the most uncertain factor for the supply of consistent good-quality fibres throughout the year. Acidic and alkaline preservation techniques seem to be the best methods, but more research is needed on these processes, with special attention paid to the observed decrease in fibre strength.
The test execution assistance of Franken BV (Goes, the Netherlands), New Holland (Zedelgem, Belgium) and Greenland Zweegers (Geldrop, the Netherlands) is appreciated. The authors also wish to thank R.J.A. Gosselink and S.J. Lips of ATO-DLO in Wageningen for their contribution of analysing wet-preserved hemp and W.J. Middelhoven of the Wageningen Agricultural University for his helpfullcomments as a microbiologist.
Next